Gene therapy is changing lives today; tomorrow, it could change millions
Ohio State scientist is studying a novel gene therapy model that could lead to treatment for widespread neurological diseases.
It was a scene that would have been unimaginable a year and a half earlier.
JuJu Stagg — unable to walk, control his muscles or speak months earlier — now helped along at the elbows by his beaming parents, standing firmly on the brick walkway in front of The Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute.
It was there that a breakthrough engineered by Krystof Bankiewicz, MD, PhD, brought new abilities to the 7-year-old. Or, as his mother, Shante Stagg, put it in a grateful email to Dr. Bankiewicz:
“Here’s a picture of JuJu standing in front of the place where you changed his life!”

Jamell and Shante Stagg with their son, JuJu, who was unable to stand and walk before he came to The Ohio State University Wexner Medical Center for a novel gene therapy surgery in 2020.
JuJu was born with an extremely rare genetic condition known as AADC deficiency. Children born with AADC lack an enzyme that produces dopamine in the central nervous system, leaving them unable to control their muscles, speak or hold up their heads.
For JuJu, it also meant seizure-like episodes known as oculogyric crises, which would go on for hours every few days.
But a new method of gene therapy pioneered by Dr. Bankiewicz, who serves as director of the Ross Center for Brain Health and Performance at Ohio State, as well as the Gilbert and Kathryn Mitchell Chair and vice chair of research at The Ohio State University College of Medicine, gave JuJu a new life.
This surgery, completed by a large and multidisciplinary neurosurgery team, delivers targeted gene therapy to the midbrain, the smallest area of the brainstem that manages several important functions in motor movement, auditory and visual processing. This targeted gene therapy is designed to help patients who are born without the ability to synthesize dopamine and serotonin, leading to a life-threatening condition that causes physical and developmental disabilities.

Dr. Bankiewicz and his colleagues now believe this gene therapy approach has the potential to be applied in the future to diseases such as Alzheimer’s and Parkinson’s disease, with some early clinical trials underway at Ohio State.
A year and a half after having the procedure at The Ohio State University Wexner Medical Center, JuJu is learning to walk and speak, according to his proud mom.
“He’s literally such a different kid,” Shante says. “It’s been really fun seeing him become his own little person. He can actually enjoy life.”
She is unequivocal in her feelings for the man who made it happen.
“Just to know what he does for such a small community — there’s only a few hundred kids in the world that have this disease — that speaks volumes to him and his heart,” she says. “He’s completely changing the lives of these children, and it is absolutely astounding.
 “My husband and I will always have a special place in our hearts for him.”
“My husband and I will always have a special place in our hearts for him.”
The Staggs were so moved by Dr. Bankiewicz, they even dressed JuJu as Dr. Bankiewicz for Halloween.
The magnitude of the breakthrough is not lost on Dr. Bankiewicz.
“I am certain that this has been the most profound experience in my whole professional career, to make such a change in people’s lives, that does not happen very often,” he says. “And that stems from the fact that I can probably see it from a different angle right now since I became a grandfather.
“Seeing the impact that we have made let me work even harder than I would normally do, seeing a chance to be able to get kids somehow restored. That really made a huge, huge impact on me.”
An early fascination with gene therapy’s potential
A native of Poland, Dr. Bankiewicz came to love science and medicine through a family friend who worked as a surgeon. This interest in clinical surgery brought him to the National Institutes of Health (NIH) in the mid-1980s, where he was awarded a fellowship in neurosurgery.
His time at the NIH was crucial to him learning how to translate research breakthroughs into clinical treatments.
“I entered this completely new world not really known to me at the time, where I was able to start asking questions in terms of how to bring the things that we do in the laboratory into the clinic,” Dr. Bankiewicz says. “And that was it, pretty much. I found my calling.”
 Among those who noticed Dr. Bankiewicz’s groundbreaking work was neurosurgeon Russell Lonser, MD, who now serves as professor and chair in the Department of Neurological Surgery and holds the Dardinger Family Endowed Chair in Oncological Neurosurgery at The College of Medicine. He remembers Dr. Bankiewicz’s passion as colleagues with similar interests in nervous system drug delivery.
Among those who noticed Dr. Bankiewicz’s groundbreaking work was neurosurgeon Russell Lonser, MD, who now serves as professor and chair in the Department of Neurological Surgery and holds the Dardinger Family Endowed Chair in Oncological Neurosurgery at The College of Medicine. He remembers Dr. Bankiewicz’s passion as colleagues with similar interests in nervous system drug delivery.
“Perseverance and tenacity are the two words that best describe him,” Dr. Lonser says. “He’s continued to move the field forward in nervous system gene therapy for over two decades. It was really the tenacity that has led to the success we see today.
“He has single-mindedly pulled the field forward.”
Dr. Bankiewicz also began studying certain models of Parkinson’s disease during his time at the NIH, work that led him to complete his PhD in neuroscience. He gained another key component of his skill set when he began working for one of the first gene therapy companies, in Alameda, California, where he learned how to develop treatments for widespread use.
The treatment for children with AADC deficiency is noteworthy in that it involves operating on a portion of the brain that was not always considered safe to operate on. During the surgery, doctors slowly infuse a benign virus programmed with specific DNA into precisely targeted areas of the middle of the brain.
A team led by Brad Elder, MD, director of Neurosurgical Oncology, performs the surgery using the virus vector that Dr. Bankiewicz pioneered. A talented team of physicians, nurses and a range of other medical professionals contribute to the work that makes these improvements possible.
Gene therapy delivery

Scientists modify the genetic code of the patient’s DNA.
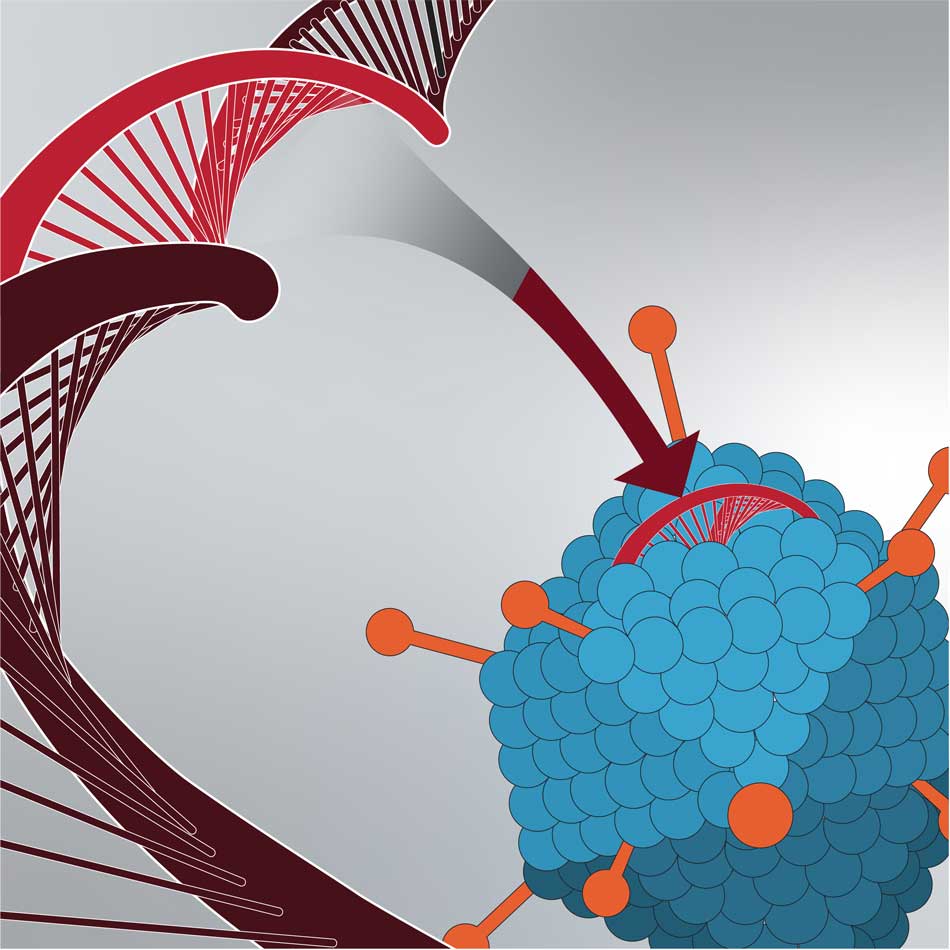
Modified DNA is injected into a vector. The vector is typically a harmless virus that serves as a vehicle to deliver the modified DNA to the cell.
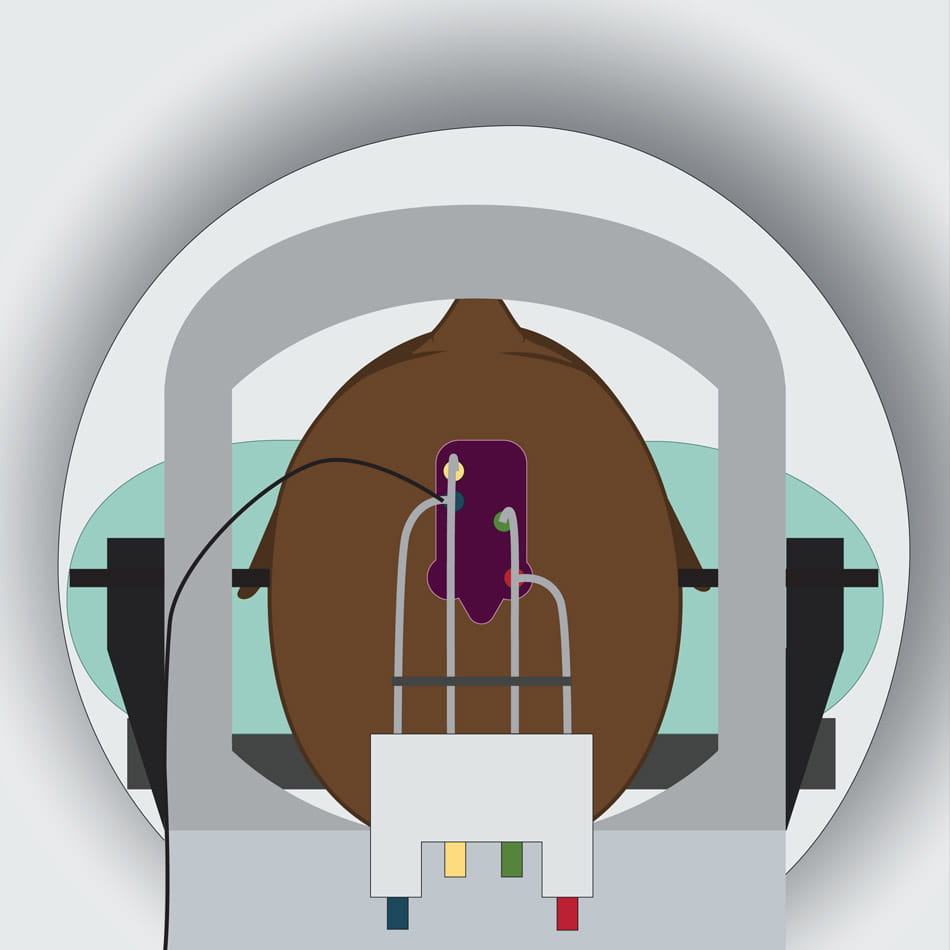
The vector is injected through the skull to a targeted cell within the brain using a delivery device.
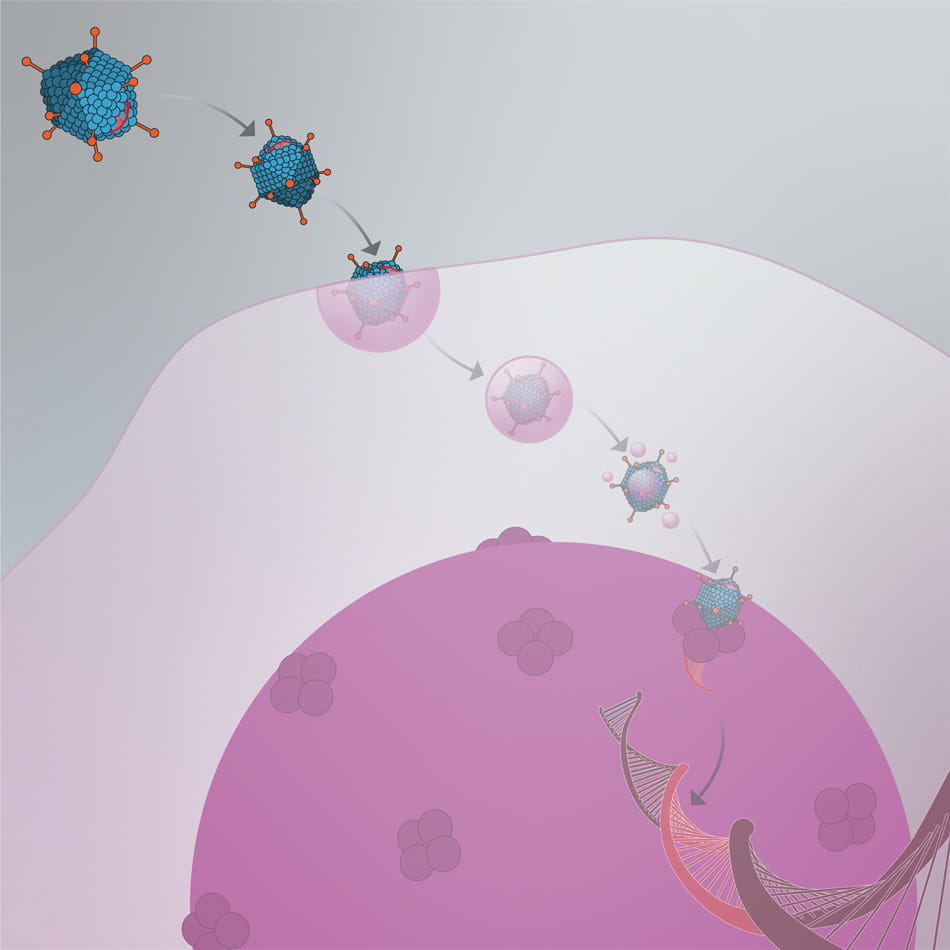
The vector attaches to the cell and releases the new gene into the cell’s nucleus.
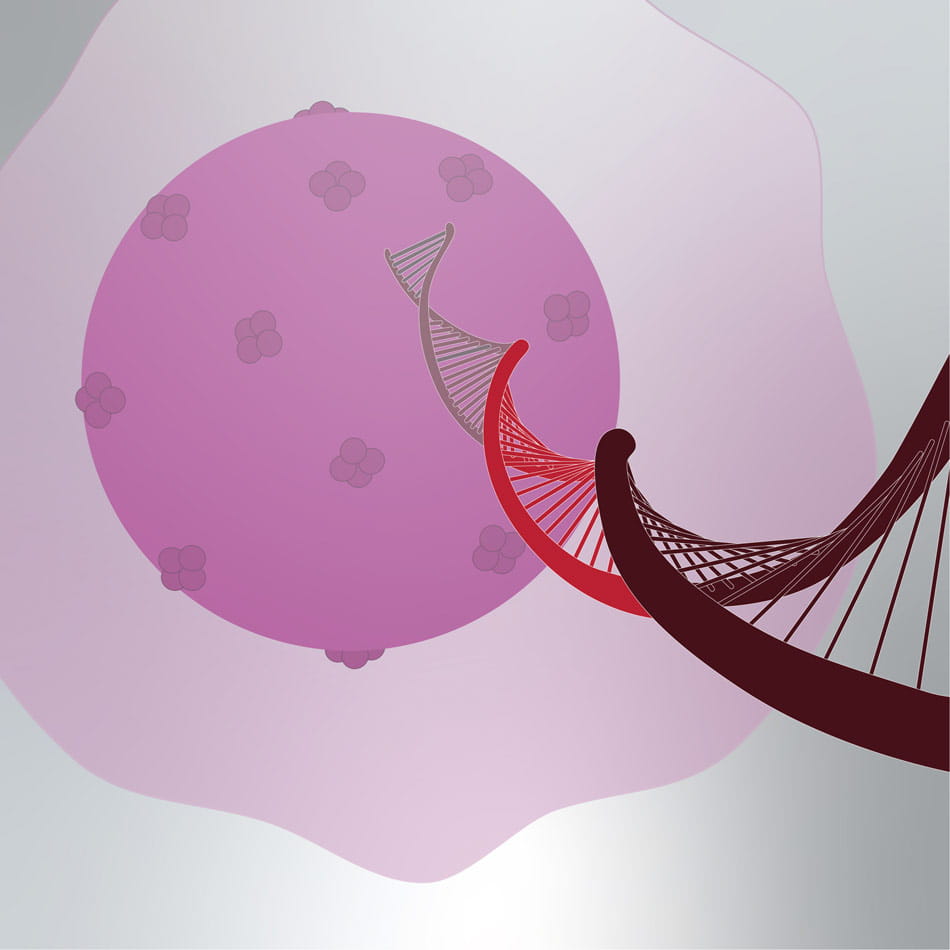
The cell makes new protein using the new gene.
Putting gene therapy into practice
Gene therapy is a technique of modifying people’s genes to treat specific diseases. This can be done by replacing a defective gene with a healthy version, deactivating a defective gene or introducing a new gene to the body. These genes are delivered via a vector, most commonly a virus that has been neutralized of its ability to cause disease. The body’s cells then take up the new genes, where they begin the work they’ve been designed to accomplish. In the case of cells within the body, such as AADC deficiency patients, the vector must be injected directly into the body.
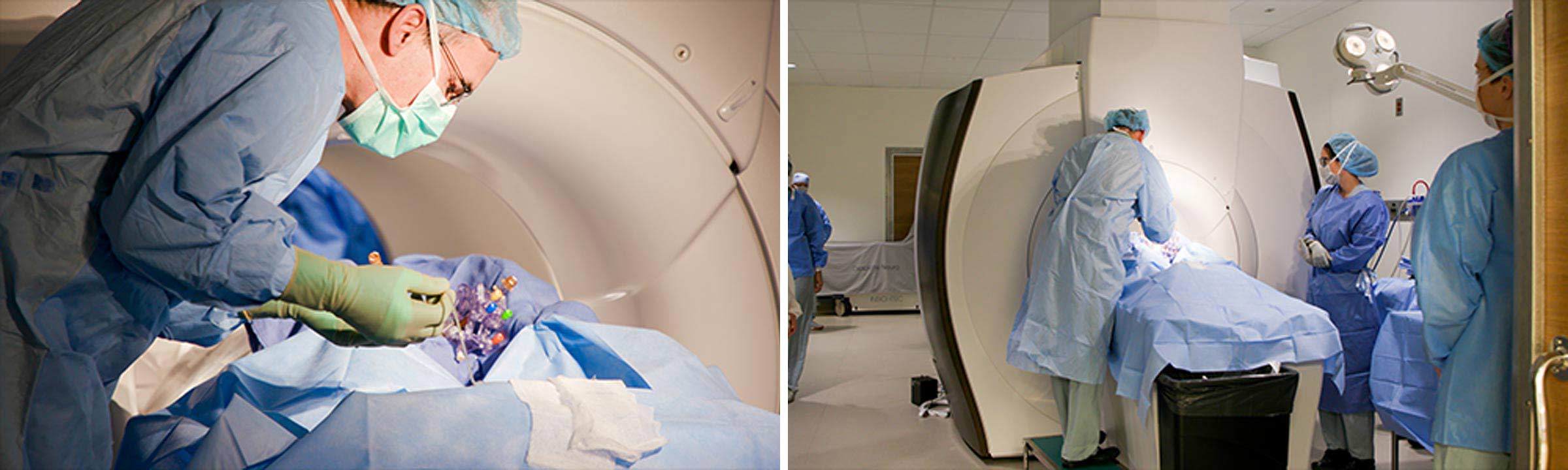
Left: Dr. Brad Elder performs gene therapy surgery on a patient at The Ohio State University Wexner Medical Center using a novel delivery vector developed by Dr. Bankiewicz. Right: Ginny Hadley, surgical tech lead for Neuro-Oncology, assists with the procedure.
Because of his previous involvement in clinical trials to treat Parkinson’s disease, Dr. Bankiewicz was certain that a similar course of treatment could be effective in children with this rare disease.
“Because of the confidence level that we have developed over the years treating patients with Parkinson’s disease with gene therapy, it allowed us to go to a rather dangerous part of the brain of those children,” he says. “Performing this type of surgery without the devices and the technology that we’ve developed in patients with Parkinson’s disease would be very difficult and dangerous.”
The breakthrough was recognized by the NIH in 2021, when it awarded a $14.6 million grant to Dr. Bankiewicz’s team to continue studying treatments for AADC deficiency. JuJu was one of 10 children treated in the United States during a previous NIH-funded trial, along with 18 in Poland. The new grant will expand that pool.
How academia — and Ohio State — made leadership in gene therapy possible
As Dr. Bankiewicz is quick to note, diseases as rare as AADC deficiency are not always priorities when it comes to research and funding. He attributes the success of this project to the work done at academic health centers.
“A lot of investment from industry, for example, that could or should have gone into those ultra-rare disorders, I think, isn’t there,” he says. “So, that’s why academia is so critical in that step, and everything I’ve done in those AADC children has been funded by the NIH or charitable foundations.
“So, I’m very grateful, of course, for the NIH to recognize the importance of developing gene therapy technology. There is no doubt in my mind that this is already impacting what we do in diseases such as Parkinson’s or Alzheimer’s. What we’ve learned is very unique and is applied now to disorders that affect millions.”
Dr. Lonser attributes the success of research projects such as Dr. Bankiewicz’s to the leadership and culture at the university.
“Ohio State’s really a phenomenal place,” Dr. Lonser says. “Both he and I have worked on both coasts throughout our careers, and I think what’s really been unique about Ohio State is that they have incredible leadership that can see the innovative pieces.”
Among those innovative pieces is the Gene Therapy Institute, which Ohio State launched in spring 2022. Led by Drs. Lonser and Bankiewicz, it will bring together related research being done in gene therapy by more than 50 faculty across the colleges of Medicine, Arts and Sciences, Law, Business, Veterinary Medicine, Engineering and Pharmacy.
 “This new institute will be a model for how collaboration can solve some of medicine’s most pressing challenges,” says Peter Mohler, PhD, vice president of research at The Ohio State University and chief scientific officer at the Ohio State Wexner Medical Center and College of Medicine. “Bringing together our experts from a broad range of disciplines will lead directly to new, life-changing treatments for patients as well as new discoveries in fundamental biology. The team-based approach to furthering gene therapy research extends outside of campus – private philanthropic support has been vital to advancing Ohio State’s work in this area as well.”
“This new institute will be a model for how collaboration can solve some of medicine’s most pressing challenges,” says Peter Mohler, PhD, vice president of research at The Ohio State University and chief scientific officer at the Ohio State Wexner Medical Center and College of Medicine. “Bringing together our experts from a broad range of disciplines will lead directly to new, life-changing treatments for patients as well as new discoveries in fundamental biology. The team-based approach to furthering gene therapy research extends outside of campus – private philanthropic support has been vital to advancing Ohio State’s work in this area as well.”

Dr. Bankiewicz’s lab team views a slide in the lab.
Applying these gene therapy techniques to widespread diseases
Dr. Lonser is most excited about the opportunity to translate these gene therapy techniques to patients with Parkinson’s, Alzheimer’s or Huntington’s disease, a rare, fatal affliction in which nerve cells in the brain break down.
At Ohio State, there are currently active trials in both Parkinson’s and Alzheimer’s disease. They are first-in-human trials taking place here. A wide range of experts from the Neurological Institute, including Dr. Elder and others, are among the teams collaborating on these studies.
“For kids with AADC deficiency, this treatment is saving their lives,” Dr. Lonser says. “If we can apply these breakthroughs to Huntington’s disease, it would be an opportunity to treat that disease for the first time. We are the largest site for gene therapy trials in the world, and we run more first in-human trials than anywhere else. Each of these is potentially offering a breakthrough in a deadly disease that’s uncurable.”
As the focus on gene therapy increases, Dr. Bankiewicz is confident that the next breakthrough to help patients like JuJu will be discovered at Ohio State.
“The last decade has brought tremendous interest in gene therapy and a lot of successes, as well,” he says. “Here at The Ohio State University, we are very interested in pursuing gene therapy. Since my arrival here, we are the only institution in the world that is performing most of the gene therapy trials in the brain.
“I think we are in a very unique position here. Clearly the future looks bright at The Ohio State University, as far as gene therapy is concerned.”
And just how big of an effect can that have on the lives of patients? How about supplanting the most important day of their lives? It’s already happened in the case of children treated by this breakthrough.
“The most profound impact I see is families that have stopped celebrating the child’s natural birthday,” Dr. Bankiewicz says. “They celebrate the date when they got the gene therapy. That’s their new birthday.”

Your support fuels life-changing gene therapy
Give to support groundbreaking gene therapy research at Ohio State.
Ways to Give
Transforming lives for people with brain and spine disorders
Learn more and schedule an appointment.
Expert care starts here



